Indole-2,3-dione(8CI);2,3-Dihydro-1H-indole-2,3-dione;2,3-Dihydroindole-2,3-dione;2,3-Diketoindoline;2,3-Dioxo-2,3-dihydroindole;2,3-Dioxoindoline;2,3-Indolindione;2,3-Indolinedione;Isatic acid lactam;Isatin;Isatine;Isatinic acid anhydride;NSC 9262;Pseudoisatin;o-Aminobenzoylformic anhydride;
 Xi,
Xi, Xn
Risk Codes:
Transport Information:
MSDS:
Xn
Risk Codes:
Transport Information:
MSDS:

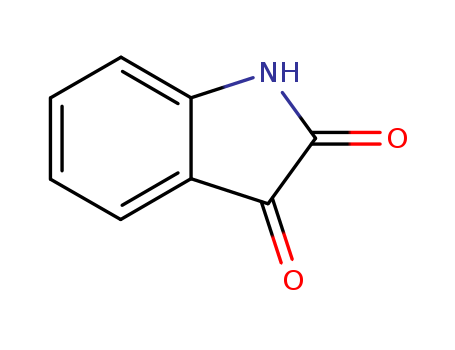
IUPAC: 1H-indole-2,3-dione
CAS:91-56-5
The Molecular formula of INDOLE-2,3-DIONE(91-56-5):C8H5NO2
The Molecular Weight of INDOLE-2,3-DIONE(91-56-5):147.13
Synonyms:Isatin;TIMTEC-BB SBB009100;O-AMINOPHENYLGLYOXALIC LACTIM;O-AMINOBENZOYLFORMIC ACID;O-AMINOBENZOYLFORMIC ANHYDRIDE;AKOS BBS-00003223;2,3-DIKETOINDOLINE;2,3-DIHYDROINDOLE-2,3-DIONE;2,3-INDOLEDIONE
EINECS:202-077-8
Density:1.367g/cm3
Melting Point:198-204oC (Decomposes)(lit.)
Boiling Point:360.3oC at 760 mmHg
Flash Point:220oC
Solubility:Slightly soluble SOLVENT AUTOIGNITION
Appearance:yellow to reddish crystalline solid
liansport Information:25kgs
Product Categories:Intermediates;Heterocyclic Compounds;Indoles;Simple Indoles
Mol File:91-56-5.mol
storage temp.:Store at RT.
Decomposition:194oC
Merck:14,5104
BRN:383659
INDOLE-2,3-DIONE(91-56-5) is an indole derivative. In 1841, Erdman and Laurent first obtained the compound as a product from the oxidation of indigo dye by nitric acid and chromic acids. The compound is found in many plants.Schiff bases of INDOLE-2,3-DIONE are investigated for their pharmaceutical properties.It was observed that it forms a blue dye if it is mixed with sulfuric acid and crude benzene. The formation of the blue indophenin was long believed to be a reaction with benzene. Victor Meyer was able to isolate the substance responsible for this reaction from benzene. This new heterocyclic compound was thiophene.
| 1. | orl-rat LDLo:5 g/kg | IJPPAZ Indian Journal of Physiology and Pharmacology. 6 (1962),145. | ||
| 2. | orl-mus LD50:300 mg/kg | NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. 55 (1959),1514. | ||
| 3. | ipr-mus LD50:563 mg/kg | PCJOAU Pharmaceutical Chemistry Journal (English Translation). Translation of KHFZAN. 15 (1981),858. |
poison by ingestion. moderately toxic by intraperitoneal route. experimental reproductive effects. when heated to decomposition it emits toxic fumes of nox. see also anhydrides.
safty informations about indole-2,3-dione(91-56-5):
hazard codes:xi,xn
risk statements:36/37/38-20/21/22
36/37/38 (irritating to the eyes ,piratory system and skin)
20/21/22(harmful by inhalation, in contact with skin and if swallowed)
safety statements:26-36-24/25
26 (in case of contact with eyes,rinse immediately with plenty of water and seek medical advice)
36 (wear suitable protective clothing)
24/25(avoid contact with skin and eyes)
wgk germany:1
ridadr:2811
rtecs:nl7873000
hazardclass:6.1(b)
packinggroup:iii
hs code:29337900