Acetaldehyde,diethyl acetal (8CI);1,1-Diethoxyethane;Acetaldehyde ethyl acetal;Diethyl acetal;Ethanal diethyl acetal;Ethylidene diethyl ether;NSC 7624;
 F,
F,  Xi
Risk Codes:
Transport Information:
MSDS:
Xi
Risk Codes:
Transport Information:
MSDS:
ACETAL is reported in EPA TSCA Inventory.
DOT Classification: 3; Label: Flammable Liquid
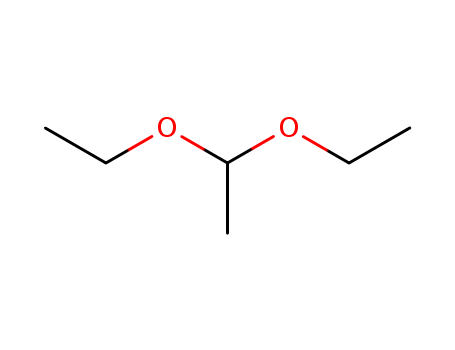
The Acetal, with the CAS registry number 105-57-7 and EINECS registry number 203-310-6, has the systematic name and IUPAC name of 1,1-diethoxyethane. It is a molecule with two single bonded oxygens attached to the same carbon atom. It is a kind of clear, colorless liquid, and belongs to the product category of Pharmaceutical Intermediates. And the molecular formula of the chemical is C6H14O2. What's more, it should be stored in the refrigerator.
It is stable compared to hemiacetals but their formation is a reversible equilibrium as with esters. And It is used as protecting groups for carbonyl groups in organic synthesis as they are stable with respect to hydrolysis by bases and with respect to many oxidizing and reducing agents. And it is also used as solvent in organic synthesis.
The physical properties of Acetal are as followings: (1)ACD/LogP: 1.15; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.14; (4)ACD/LogD (pH 7.4): 1.14; (5)ACD/BCF (pH 5.5): 4.37; (6)ACD/BCF (pH 7.4): 4.37; (7)ACD/KOC (pH 5.5): 99.95; (8)ACD/KOC (pH 7.4): 99.95; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.389; (14)Molar Refractivity: 33.29 cm3; (15)Molar Volume: 140.6 cm3; (16)Polarizability: 13.19×10-24cm3; (17)Surface Tension: 22.8 dyne/cm; (18)Density: 0.84 g/cm3; (19)Enthalpy of Vaporization: 32.85 kJ/mol; (20)Boiling Point: 103.6 °C at 760 mmHg; (21)Vapour Pressure: 37.1 mmHg at 25°C.
Preparation: This chemical can be prepared by Ethanol and acetaldehyde. The reaction will need catalyst anhydrous calcium chloride.
![]()
![]()
You should be cautious while dealing with this chemical. It is a kind of highly flammable chemcial which irritates to eyes and skin. Therefore, you had better take the following instructions: Keep container in a well-ventilated place; Keep away from sources of ignition - No smoking; Take precautionary measures against static discharges.
You can still convert the following datas into molecular structure:
(1)SMILES: CCOC(C)OCC
(2)InChI: InChI=1S/C6H14O2/c1-4-7-6(3)8-5-2/h6H,4-5H2,1-3H3
(3)InChIKey: DHKHKXVYLBGOIT-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 500mg/kg (500mg/kg) | National Technical Information Service. Vol. AD277-689, | |
| mouse | LD50 | oral | 3500mg/kg (3500mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 42(3), Pg. 12, 1977. | |
| rabbit | LD50 | oral | 3545mg/kg (3545mg/kg) | Proceedings of the Society for Experimental Biology and Medicine. Vol. 29, Pg. 730, 1932. | |
| rat | LCLo | inhalation | 4000ppm/4H (4000ppm) | Journal of Industrial Hygiene and Toxicology. Vol. 31, Pg. 343, 1949. | |
| rat | LD50 | intraperitoneal | 900mg/kg (900mg/kg) | "Industrial Hygiene and Toxicology," 2nd ed., Patty, F.A., ed., New York, John Wiley & Sons, Inc., 1958-63Vol. 2, Pg. 1982, 1963. | |
| rat | LD50 | oral | 4600mg/kg (4600mg/kg) | Medizin und Ernaehrung. Vol. 8, Pg. 244, 1967. |