HDA;Hydroxyamine; Telclean 1200A

 Risk Codes:
Transport Information:
MSDS:
Risk Codes:
Transport Information:
MSDS:
Chemistry informtion about Hydroxylamine (CAS NO.7803-49-8) is:
IUPAC Name: Hydroxylamine
Synonyms: Hydroxylamine,Freebase ; Hydroxylaminefree-Base ; Nh2oh ; Oxammonium ; Oxyammonia ; Fh-50 ; Fh-50tm ; Hydroxylamine
MF: H3NO
MW: 33.03
EINECS: 232-259-2
Melting Point: 7°C
Density: 1.111 g/cm3
Boiling Point: 56.5 °C at 760 mmHg
Vapour Pressure: 179 mmHg at 25°C
Enthalpy of Vaporization: 34.9 kJ/mo
Refractive Index: n20/D 1.393
Merck: 13,4853
Stability: Stable. Incompatible with oxidizing agents, potassium dichromate, chromium trioxide, zinc, calcium, copper, sodium, ammonia, carbonyls, phosphorus halides, pyridine, hypochlorites.
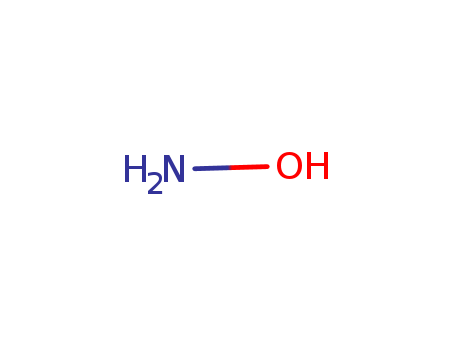
Following is the molecular structure of Hydroxylamine (CAS NO.7803-49-8) is:
![]()
Hydroxylamine (CAS NO.7803-49-8) and its salts are commonly used as reducing agents in a myriad of organic and inorganic reactions. They can also act as antioxidants for fatty acids. Some non-chemical uses include removal of hair from animal hides and photography developing solutions. It can also be used to highly selectively cleave asparaginyl-glycine peptide bonds in peptides and proteins. It also bonds to and permanently disables (poisons) heme-containing enzymes. It is used as an irreversible inhibitor of the oxygen-evolving complex of photosynthesis on account of its similar structure to water.
NH2OH can be synthesized via several routes.
The first route is via Raschig synthesis: aqueous ammonium nitrite is reduced by HSO4− and SO2 at 0°C to yield a hydroxylamido-N,N-disulfate anion:
NH4NO2 + 2 SO2 + NH3 + H2O → 2 NH4+ + N(OH)(OSO2)22−
This anion is then hydrolyzed to give (NH3OH)2SO4:
N(OH)(OSO2)22− + H2O → NH(OH)(OSO2)− + HSO4−
2 NH(OH)(OSO2)− + 2 H2O → (NH3OH)2SO4
Solid NH2OH can be collected by treatment with liquid ammonia. Ammonium sulfate, (NH4)2SO4, a side-product insoluble in liquid ammonia, is removed by filtration; the liquid ammonia is evaporated to give the desired product.Another route is to make hydroxylammonium salts which can then be converted to hydroxylamine.
(NH3OH)Cl + NaOBu → NH2OH + NaCl + BuOH
Hydroxylamine can also be produced by the reduction of nitrous acid or potassium nitrate with bisulfite:
HNO2 + 2 HSO3− → N(OH)(OSO2)22− + H2O → NH(OH)(OSO2)− + HSO4−
NH(OH)(OSO2)− + H3O+ (100 °C/1 h) → NH3(OH)+ + HSO4−
| 1. | mmo-omi 1 mg/L | MUREAV Mutation Research. 74 (1980),113. | ||
| 2. | slt-dmg-unr 45 mmol/L/48H | MUREAV Mutation Research. 120 (1983),233. | ||
| 3. | dni-hmn:hla 10 mmol/L | MUREAV Mutation Research. 93 (1982),447. | ||
| 4. | sce-ham:lng 5 mmol/L | HUGEDQ Human Genetics. 54 (1980),155. | ||
| 5. | ipr-rat LD50:59 mg/kg | CNREA8 Cancer Research. 26 (1966),1448. | ||
| 6. | scu-rat LD50:29 mg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 119 (1957),444. | ||
| 7. | ipr-mus LD50:60 mg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 165 (1969),30. |
Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
a poison by intraperitoneal and subcutaneous routes. a corrosive irritant to the eyes, skin, and mucous membranes. locally it is irritating, and systemically it can cause methemoglobinemia. human mutation data reported. dangerous fire hazard when exposed to heat, flame, and oxidizers. may ignite spontaneously in air if a large surface area is exposed (e.g., precipitate on paper). explodes in air when heated above 70°c. explosive reaction with potassium dichromate, chromium trioxide, powdered zinc + heat. forms the heat-sensitive explosive bis(hydroxylamide) in reaction with zinc or calcium. ignites on contact with copper(ii) sulfate, metals (e.g., sodium), oxidants (e.g., barium peroxide, barium oxide, lead dioxide, potassium permanganate, chlorine), phosphorus chlorides (e.g., phosphorus trichloride, phosphorus pentachloride). incompatible with carbonyls, pyridine. vigorous reaction with hypochlorites. when heated to decomposition it emits toxic fumes of nox. see also amines.
hazard codes:
xn: harmful![]()
n: dangerous for the environment![]()
risk statements:
r5: heating may cause an explosion.
r22: harmful if swallowed.
r37/38: irritating to respiratory system and skin.
r41: risk of serious damage to the eyes.
r43: may cause sensitization by skin contact.
r48/22: harmful: danger of serious damage to health by prolonged exposure if swallowed.
r50: very toxic to aquatic organisms.
safety statements:
s23: do not breathe vapour.
s26: in case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
s36/37/39: wear suitable protective clothing, gloves and eye/face protection.
s47: keep at temperature not exceeding...
s61: avoid release to the environment. refer to special instructions / safety data sheets.
s22: do not breathe dust.
ridadr: un 3082 9/pg 3
wgk germany: 3
f:
f 4.8: do not heat over 80°c.
hazardclass: 8
packinggroup: ii
General description about Hydroxylamine (CAS NO.7803-49-8) ,it is a odorless white crystalline solid. Sinks and mixes with water.
Air & Water Reactions: Decomposes rapidly at room temperature or when dissolved in hot water by internal oxidation-reduction. Reacts with water or steam to produce heat and corrosive liquids.
Reactivity Profile: It is a white solid, thermally unstable, decomposes rapidly at room temperature or when dissolved in hot water by internal oxidation-reduction. It should be stored below 10° C [Bailar, 1973, vol. 2, p. 272]. Explosive reaction with strong oxidizers (chromium trioxide, potassium dichromate) or powdered zinc upon heat. Reaction with zinc or calcium produces explosive bishydroxylamides. It ignites on contact with cupric sulfate, alkali metals (sodium, potassium), oxidants (e.g., barium oxide, barium peroxide, lead dioxide, potassium permanganate, chlorine), phosphorus trichloride and pentachloride. It reacts vigorously with hypochlorites, pyridine, carbonyls [Sax, 9th ed., 1996, p. 1875]. On contact with organic materials in thin layer (e.g., crystals on filter paper), it may ignite spontaneously in air. It explodes when heated above 70° C [Brauer, 1963, vol. 1, p. 502]. During a distillation process, an explosion occurred. Potassium hydroxide is thought to be involved in the explosion. Employees in the plant complained of chest pains and suffered chemical burns. Five people were killed by the explosion.
Health Hazard: Inhalation: Moderately toxic by inhalation and oral routes with the following symptoms possible: headache, vertigo, tinnitus, dyspnea, nausea and vomiting, cyanosis, proteinuria and hematuria, jaundice, restlessness, and convulsion. Methemoglobinemia has been reported. Eyes: Corrosive - highly irritating. Skin: Irritating or corrosive to skin. Ingestion: Moderately toxic by inhalation and oral routes with the following symptoms possible; headache, vertigo, tinnitus, dyspnea, nausea and vomiting, cyanosis, proteinuria and hematuria, jaundice, restlessness, and convulsion. Methemoglobinemia has been reported.