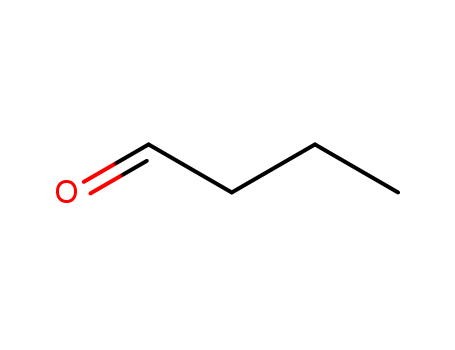
Butyraldehyde(8CI);Butal;Butaldehyde;Butanaldehyde;Butyl aldehyde;Butyral;Butyricaldehyde;Butyrylaldehyde;NSC 62779;n-Butanal;n-Butyl aldehyde;n-Butyraldehyde;n-Butyric aldehyde;Butanal;
 F
Risk Codes:
Transport Information:
MSDS:
F
Risk Codes:
Transport Information:
MSDS:
The Butanal, with the CAS registry number 123-72-8 and EINECS registry number 204-646-6, is the aldehyde derivative of butane. It is a colourless flammable liquid with an acrid smell, and miscible with most organic solvents. The molecular formula of this chemical is C4H8O.
The physical properties of Butanal are as followings: (1)ACD/LogP: 0.91; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.91; (4)ACD/LogD (pH 7.4): 0.91; (5)ACD/BCF (pH 5.5): 2.88; (6)ACD/BCF (pH 7.4): 2.88; (7)ACD/KOC (pH 5.5): 74.13; (8)ACD/KOC (pH 7.4): 74.13; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 17.07 Å2; (13)Index of Refraction: 1.369; (14)Molar Refractivity: 20.76 cm3; (15)Molar Volume: 91.8 cm3; (16)Polarizability: 8.23×10-24cm3; (17)Surface Tension: 22.5 dyne/cm; (18)Density: 0.784 g/cm3; (19)Enthalpy of Vaporization: 31.86 kJ/mol; (20)Boiling Point: 77.6 °C at 760 mmHg; (21)Vapour Pressure: 96 mmHg at 25°C.
Preparation of Butanal: It can be prepared by the catalytic dehydrogenation of n-butanol. And at one time, it was produced industrially by the catalytic hydrogenation of crotonaldehyde, which is derived from acetaldehyde. It is produced almost exclusively by the hydroformylation of propylene
CH3CH=CH2 + H2 + CO → CH3CH2CH2CHO
Uses of Butanal: It is an important intermediate used in plasticizers, synthetic resins, rubber accelerators and pesticides. It is also a kind of important chemical raw material which is used for the preparation of flavor & aromas.
You should be cautious while dealing with this chemical. It is a kind of highly flammable chemical. Therefore, you had better take the following instructions: Keep container in a well-ventilated place; Do not keep the container sealed; Take precautionary measures against static discharges.
You can still convert the following datas into molecular structure:
(1)SMILES: O=CCCC
(2)InChI: InChI=1/C4H8O/c1-2-3-4-5/h4H,2-3H2,1H3
(3)InChIKey: ZTQSAGDEMFDKMZ-UHFFFAOYAZ
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mammal (species unspecified) | LC50 | inhalation | 38gm/m3 (38000mg/m3) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 51(5), Pg. 61, 1986. | |
| mouse | LC50 | inhalation | 44610mg/m3/2H (44610mg/m3) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 30, 1982. | |
| mouse | LD50 | intraperitoneal | 1140mg/kg (1140mg/kg) | Food and Cosmetics Toxicology. Vol. 17, Pg. 731, 1979. | |
| mouse | LD50 | subcutaneous | 2700mg/kg (2700mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC KIDNEY, URETER, AND BLADDER: HEMATURIA | Acta Pharmacologica et Toxicologica. Vol. 6, Pg. 299, 1950. |
| rabbit | LD50 | skin | 3560uL/kg (3.56mL/kg) | Union Carbide Data Sheet. Vol. 7/20/1965, | |
| rat | LCLo | inhalation | 8000ppm/4H (8000ppm) | National Technical Information Service. Vol. OTS0516688, | |
| rat | LD50 | intraperitoneal | 800mg/kg (800mg/kg) | Food and Cosmetics Toxicology. Vol. 17, Pg. 731, 1979. | |
| rat | LD50 | oral | 2490mg/kg (2490mg/kg) | National Technical Information Service. Vol. OTS0516688, | |
| rat | LD50 | subcutaneous | 10gm/kg (10000mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC | Acta Pharmacologica et Toxicologica. Vol. 6, Pg. 299, 1950. |