Benzeneacetic acid, .alpha.-methyl-4- (2-methylpropyl)-;2-(4-Isobutylphenyl)propanoic acid;(.+ -.)-2-(p-Isobutylphenyl)propionic acid;Benzeneacetic acid,R-methyl-4-(2-methylpropyl)-;2-(4-Isobutylphenyl)propionic Acid;Motrin;(2RS)-2-[4-(2-methylpropyl)phenyl]propanoic acid;(4-Isobutylphenyl)-alpha-methylacetic acid;(+-)-ibuprofen;(+-)-alpha-Methyl-4-(2-methylpropyl)benzeneacetic acid;
 Xn,
Xn, N
Risk Codes:
Transport Information:
MSDS:
N
Risk Codes:
Transport Information:
MSDS:
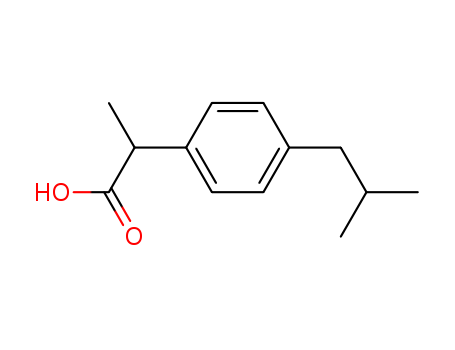
IUPAC Name: 2-[4-(2-Methylpropyl)phenyl]propanoic acid
Molecular Formula: C13H18O2
Molecular Weight: 206.28
Molecular Structure: 
H bond acceptors: 2
H bond donors: 1
Freely Rotating Bonds: 4
Polar Surface Area: 26.3 Å2
Index of Refraction: 1.518
Molar Refractivity: 60.77 cm3
Molar Volume: 200.3 cm3
Surface Tension: 38 dyne/cm
Density: 1.029 g/cm3
Flash Point: 216.7 °C
Melting Point: 77-78 °C(lit.)
Enthalpy of Vaporization: 59.25 kJ/mol
Boiling Point: 319.6 °C at 760 mmHg
Vapour Pressure: 0.000139 mmHg at 25°C
Appearance: Colourless, Crystalline Solid
Water Solubility: insoluble
Stability: Stable. Combustible. Incompatible with strong oxidizing agents
Appearance: Colourless, Crystalline Solid
SMILES: O=C(O)C(c1ccc(cc1)CC(C)C)C
InChI: InChI=1/C13H18O2/c1-9(2)8-11-4-6-12(7-5-11)10(3)13(14)15/h4-7,9-10H,8H2,1-3H3,(H,14,15)
InChIKey: HEFNNWSXXWATRW-UHFFFAOYAB
Std. InChI: InChI=1S/C13H18O2/c1-9(2)8-11-4-6-12(7-5-11)10(3)13(14)15/h4-7,9-10H,8H2,1-3H3,(H,14,15)
Std. InChIKey: HEFNNWSXXWATRW-UHFFFAOYSA-N
Product Categories of Ibuprofen (CAS NO.15687-27-1): Active Pharmaceutical Ingredients;API;APIs;Inhibitors;Intermediates & Fine
Chemicals;Pharmaceuticals;API's;Lipid signaling
Ibuprofen was derived from propionic acid by the research arm of Boots Group during the 1960s. The drug was launched as a treatment for rheumatoid arthritis in the United Kingdom in 1969, and in the United States in 1974. Famously, it is recorded that Dr. Adams initially tested his drug on a hangover. He was subsequently awarded an OBE in 1987.
Ibuprofen (CAS NO.15687-27-1) can be available in topical gel form which is absorbed through the skin, and can be used for sports injuries, with less risk of digestive problems. Ibuprofen is sometimes used for the treatment of acne, because of its anti-inflammatory properties.
Ibuprofen(15687-27-1) can be synthesized from isobutylbenzene by a Friedel-Crafts acylation with acetyl chloride, followed by formation of a cyanohydrin. Treatment with hydrogen iodide and phosphorus reduces the benzylic hydroxyl to a hydrogen and hydrolyzes the nitrile to a carboxylic acid.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| child | LDLo | oral | 469mg/kg (469mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Annals of Emergency Medicine. Vol. 15, Pg. 1308, 1986. |
| child | TDLo | oral | 480mg/kg/17D- (480mg/kg) | LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), DIFFUSE" | Journal of Pediatrics. Vol. 90, Pg. 657, 1977. |
| child | TDLo | oral | 500mg/kg (500mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" | Annals of Pharmacotherpy. Vol. 31, Pg. 432, 1997. |
| guinea pig | LD50 | oral | 495mg/kg (495mg/kg) | Archives of Toxicology, Supplement. Vol. 7, Pg. 365, 1984. | |
| hamster | LD50 | oral | 1690mg/kg (1690mg/kg) | Archives of Toxicology, Supplement. Vol. 7, Pg. 365, 1984. | |
| mammal (species unspecified) | LD50 | oral | 1gm/kg (1000mg/kg) | Acta Poloniae Pharmaceutica. For English translation, see APPFAR. Vol. 42, Pg. 545, 1985. | |
| man | LDLo | oral | 171mg/kg (171mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC VASCULAR: BP LOWERING NOT CHARACTERIZED IN AUTONOMIC SECTION | British Medical Journal. Vol. 281, Pg. 1458, 1980. |
| man | TDLo | oral | 120mg/kg/W-I (120mg/kg) | SENSE ORGANS AND SPECIAL SENSES: OTHER: EYE SKIN AND APPENDAGES (SKIN): "DERMATITIS, OTHER: AFTER SYSTEMIC EXPOSURE" | New York State Journal of Medicine. Vol. 78, Pg. 1239, 1978. |
| man | TDLo | oral | 180mg/kg/3W-I (180mg/kg) | LIVER: "JAUNDICE, CHOLESTATIC" | American Journal of Gastroenterology. Vol. 91, Pg. 1626, 1996. |
| man | TDLo | oral | 429mg/kg (429mg/kg) | LUNGS, THORAX, OR RESPIRATION: RESPIRATORY OBSTRUCTION KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" | Journal of Toxicology, Clinical Toxicology. Vol. 32, Pg. 315, 1994. |
| man | TDLo | oral | 800mg/kg (800mg/kg) | BEHAVIORAL: COMA BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Postgraduate Medical Journal. Vol. 69, Pg. 575, 1993. |
| man | TDLo | oral | 1028mg/kg (1028mg/kg) | KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" | American Journal of Emergency Medicine. Vol. 13, Pg. 375, 1995. |
| mouse | LD50 | intraperitoneal | 320mg/kg (320mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) GASTROINTESTINAL: ULCERATION OR BLEEDING FROM STOMACH | Toxicology and Applied Pharmacology. Vol. 15, Pg. 310, 1969. Link to PubMed |
| mouse | LD50 | oral | 740mg/kg (740mg/kg) | BEHAVIORAL: ANALGESIA | Pharmaceutical Chemistry Journal Vol. 14, Pg. 119, 1980. |
| mouse | LD50 | rectal | 620mg/kg (620mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) GASTROINTESTINAL: "HYPERMOTILITY, DIARRHEA" BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) | Oyo Yakuri. Pharmacometrics. Vol. 24, Pg. 415, 1982. |
| mouse | LD50 | subcutaneous | 395mg/kg (395mg/kg) | Oyo Yakuri. Pharmacometrics. Vol. 24, Pg. 415, 1982. | |
| rabbit | LD50 | oral | 1400mg/kg (1400mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 32, Pg. 1032, 1990. | |
| rabbit | LD50 | rectal | 830mg/kg (830mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 32, Pg. 1032, 1990. | |
| rat | LD50 | intraperitoneal | 626mg/kg (626mg/kg) | BEHAVIORAL: ANALGESIA | Arzneimittel-Forschung. Drug Research. Vol. 27, Pg. 1006, 1977. |
| rat | LD50 | oral | 636mg/kg (636mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 34, Pg. 280, 1984. | |
| rat | LD50 | rectal | 530mg/kg (530mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) GASTROINTESTINAL: "HYPERMOTILITY, DIARRHEA" BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) | Oyo Yakuri. Pharmacometrics. Vol. 24, Pg. 415, 1982. |
| rat | LD50 | subcutaneous | 740mg/kg (740mg/kg) | Oyo Yakuri. Pharmacometrics. Vol. 24, Pg. 415, 1982. | |
| women | TDLo | oral | 8mg/kg (8mg/kg) | BEHAVIORAL: HEADACHE | JAMA, Journal of the American Medical Association. Vol. 239, Pg. 1062, 1978. |
| women | TDLo | oral | 132mg/kg/6D-I (132mg/kg) | BLOOD: THROMBOCYTOPENIA | British Journal of Clinical Practice. Vol. 48, Pg. 51, 1994. |
| women | TDLo | unreported | 96mg/kg/3D-I (96mg/kg) | SENSE ORGANS AND SPECIAL SENSES: DIPLOPIA: EYE | JAMA, Journal of the American Medical Association. Vol. 248, Pg |
hazard codes:  xn,
xn, n
n
risk statements: 22-63-51/53
r22: harmful if swallowed
r63: possible risk of harm to the unborn child
r51/53: toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
safety statements: 61-36/37
s61: avoid release to the environment. refer to special instructions safety data sheet
s36/37: wear suitable protective clothing and gloves
ridadr: 2811
wgk germany: 3
rtecs: mu6640000
hazardclass: 6.1(b)
packinggroup: iii
Ibuprofen (CAS NO.15687-27-1), its Synonyms are (+-)-2-(p-Isobutylphenyl)propionic acid ; (+-)-Ibuprofen ; (+-)-alpha-Methyl-4-(2-methylpropyl)benzeneacetic acid ; (+-)-p-Isobutylhydratropic acid ; (4-Isobutylphenyl)-alpha-methylacetic acid ; Acide (isobutyl-4-phenyl)-2 propionique ; Benzeneacetic acid, alpha-methyl-4-(2-methylpropyl)- ; p-Isobutyl-2-phenylpropionic acid .